  1:39 write formulae for compounds formed between the ions listed in 1:38. 
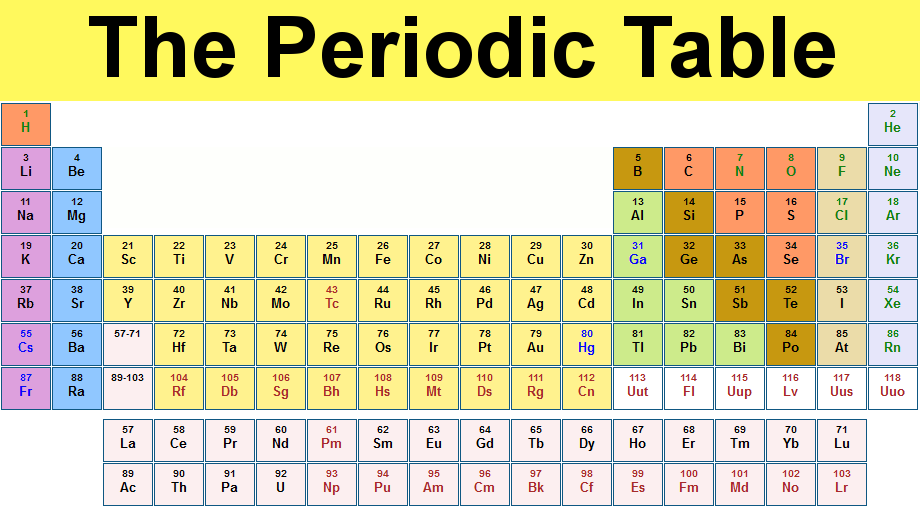
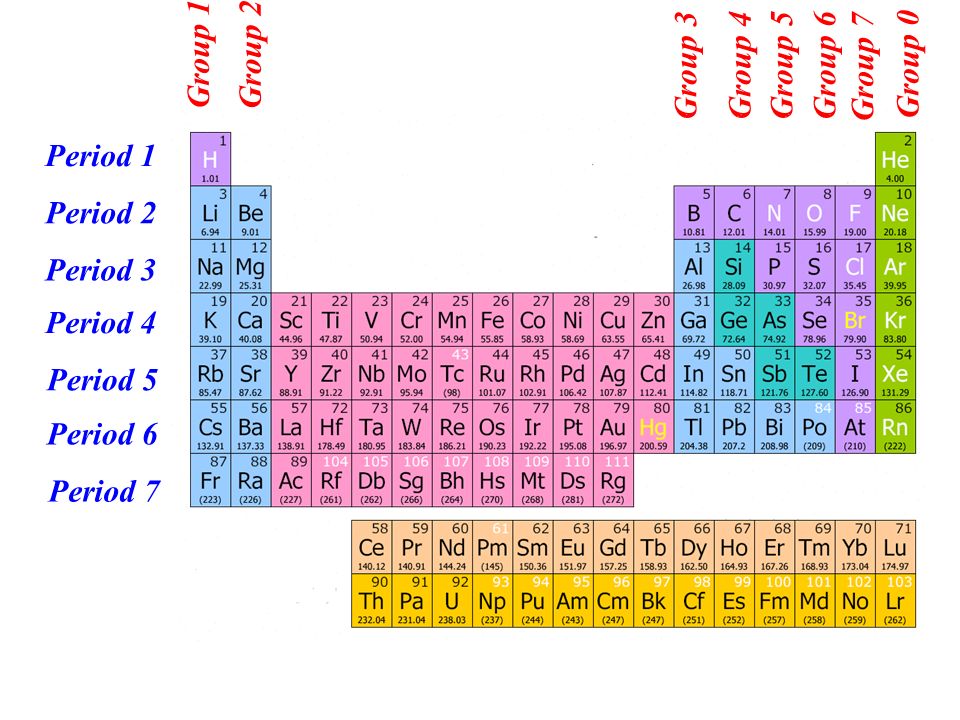
1:38 know the charges of these ions: metals in Groups 1, 2 and 3, non-metals in Groups 5, 6 and 7, Ag⁺, Cu²⁺, Fe²⁺, Fe³⁺, Pb²⁺, Zn²⁺, hydrogen (H⁺), hydroxide (OH⁻), ammonium (NH₄⁺), carbonate (CO₃²⁻), nitrate (NO₃⁻), sulfate (SO₄²⁻).1:37 understand how ions are formed by electron loss or gain. #Atomic table how to
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
